
Two sources of nitrogen-15 are the positron emission of oxygen-15 and the beta decay of carbon-15. 
Nitrogen-15 is composed of 7 protons, 8 neutrons, and 7 electrons. Nitrogen-14 is one of the very few stable nuclides with both an odd number of protons and of neutrons (seven each) and is the only one to make up a majority of its element. Nitrogen-14 is composed of 7 protons, 7 neutrons, and 7 electrons. 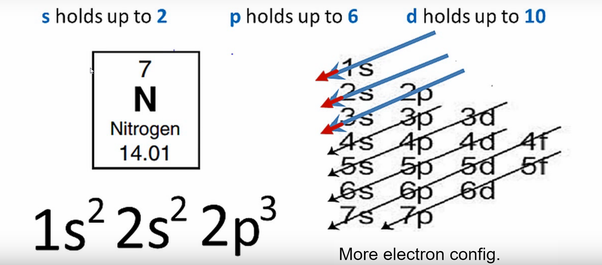
This leads to an atomic weight of around 14.007 u. The first is much more common, making up 99.634% of natural nitrogen, and the second (which is slightly heavier) makes up the remaining 0.366%. Nitrogen has two stable isotopes: 14N and 15N. Mass numbers of typical isotopes of Nitrogen are 14 15. Isotopes are nuclides that have the same atomic number and are therefore the same element, but differ in the number of neutrons. The difference between the neutron number and the atomic number is known as the neutron excess: D = N – Z = A – 2Z.įor stable elements, there is usually a variety of stable isotopes. Neutron number plus atomic number equals atomic mass number: N+Z=A. The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol N. The total electrical charge of the nucleus is therefore +Ze, where e (elementary charge) equals to 1,602 x 10 -19 coulombs. Total number of protons in the nucleus is called the atomic number of the atom and is given the symbol Z. Nitrogen is a chemical element with atomic number 7 which means there are 7 protons in its nucleus.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
